Cerene® Endometrial Cryotherapy
Provide lasting relief from her heavy, painful periods with confidence.
Say Hello to Cerene
Cerene is a well-tolerated and effective endometrial cryotherapy treatment for heavy menstrual bleeding and dysmenorrhea that preserves cavity access and her future diagnostic options. For you, it’s an efficient solution designed to fit into your practice routine - it can be performed at any site of service.
HMB &
Dysmenorrhea Relief
A well-tolerated treatment that can significantly
reduce both heavy menstrual bleeding and dysmenorrhea, delivering high satisfaction and improved quality of life
Efficient Treatment
A 2 ½ minute treatment designed to fit into your practice routine with no general anesthesia, no incisions and no capital equipment
Preserved Cavity Access
Minimal intrauterine adhesions post-cryotherapy aids in the ability to perform future diagnostic evaluations and avoid the need for a diagnostic hysterectomy
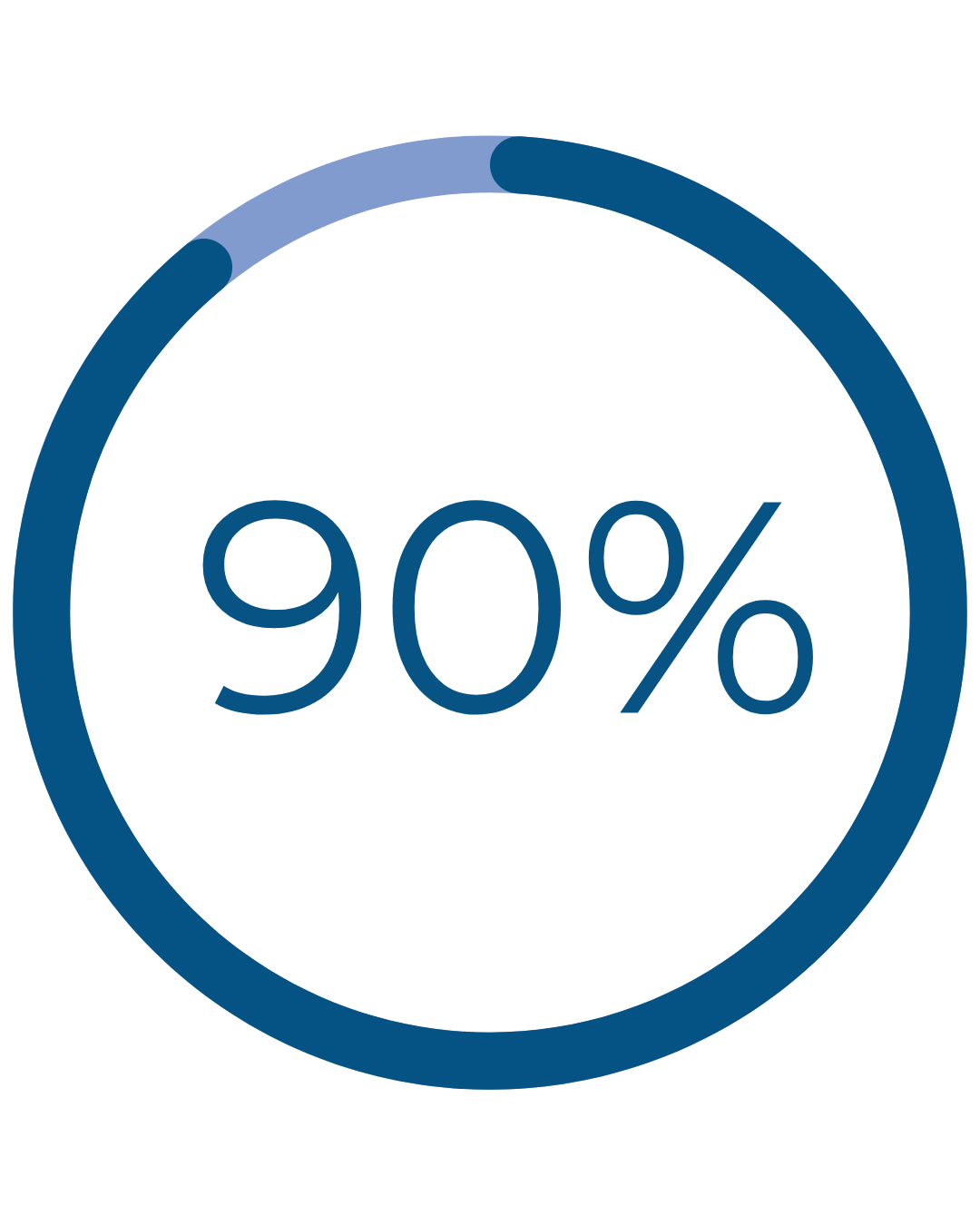
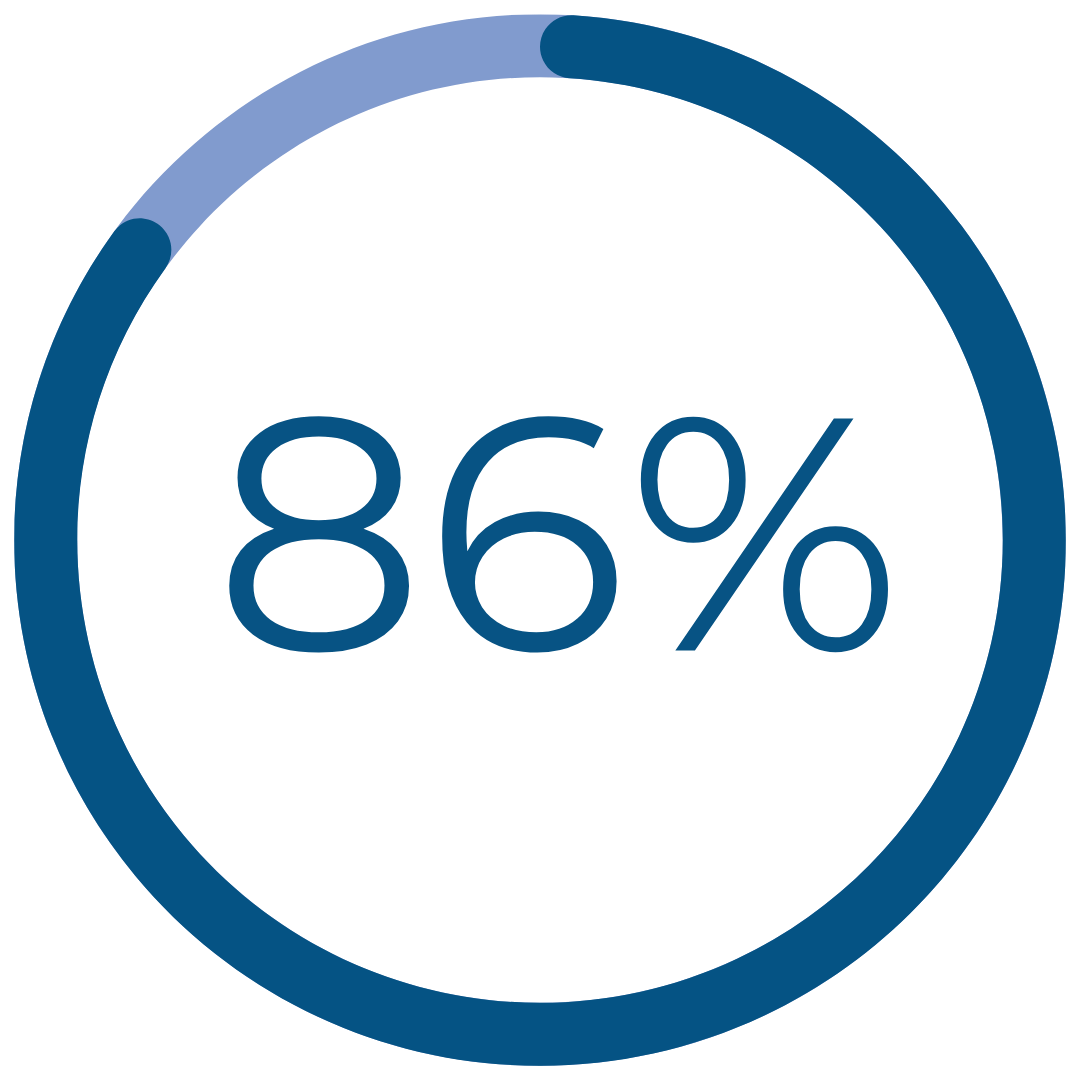
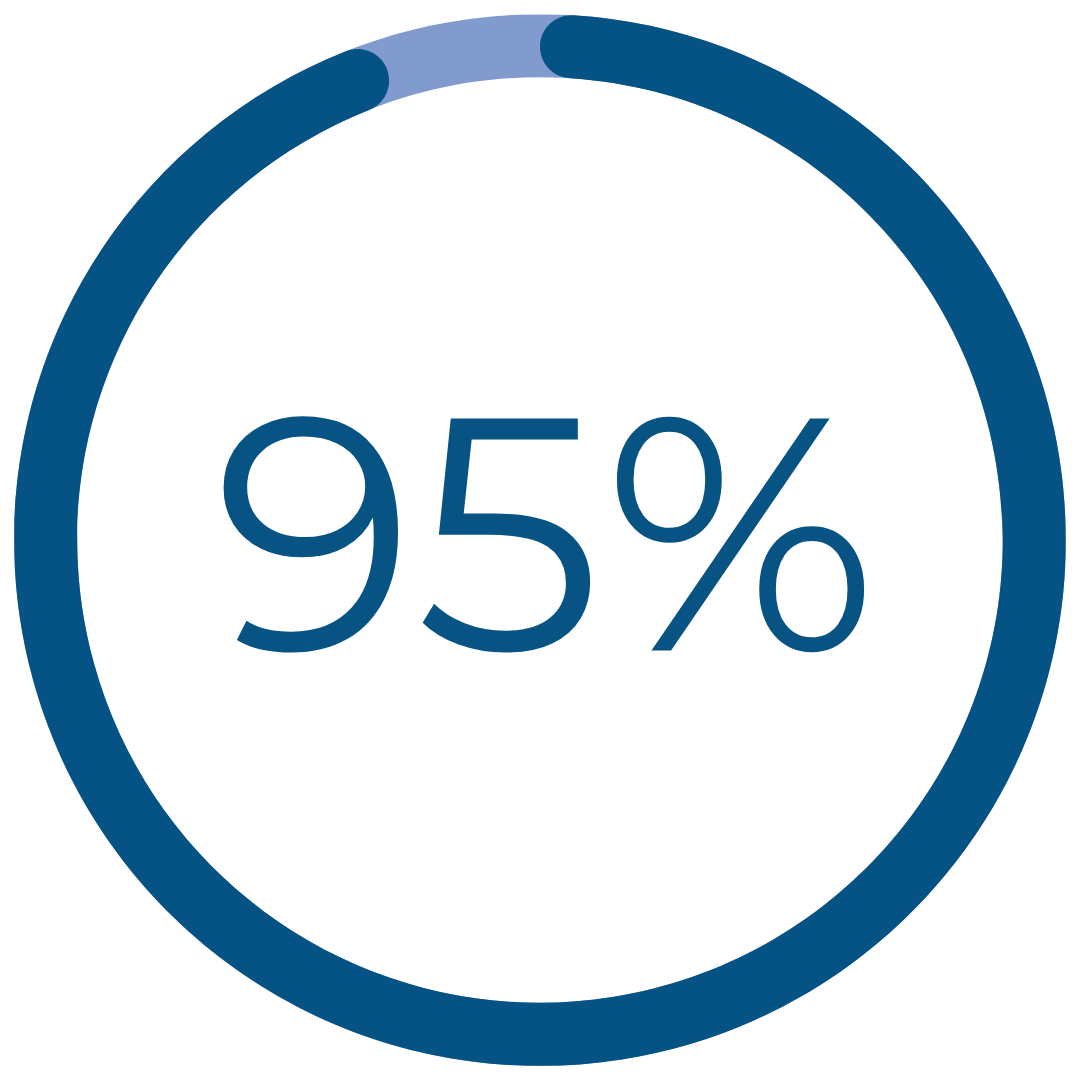
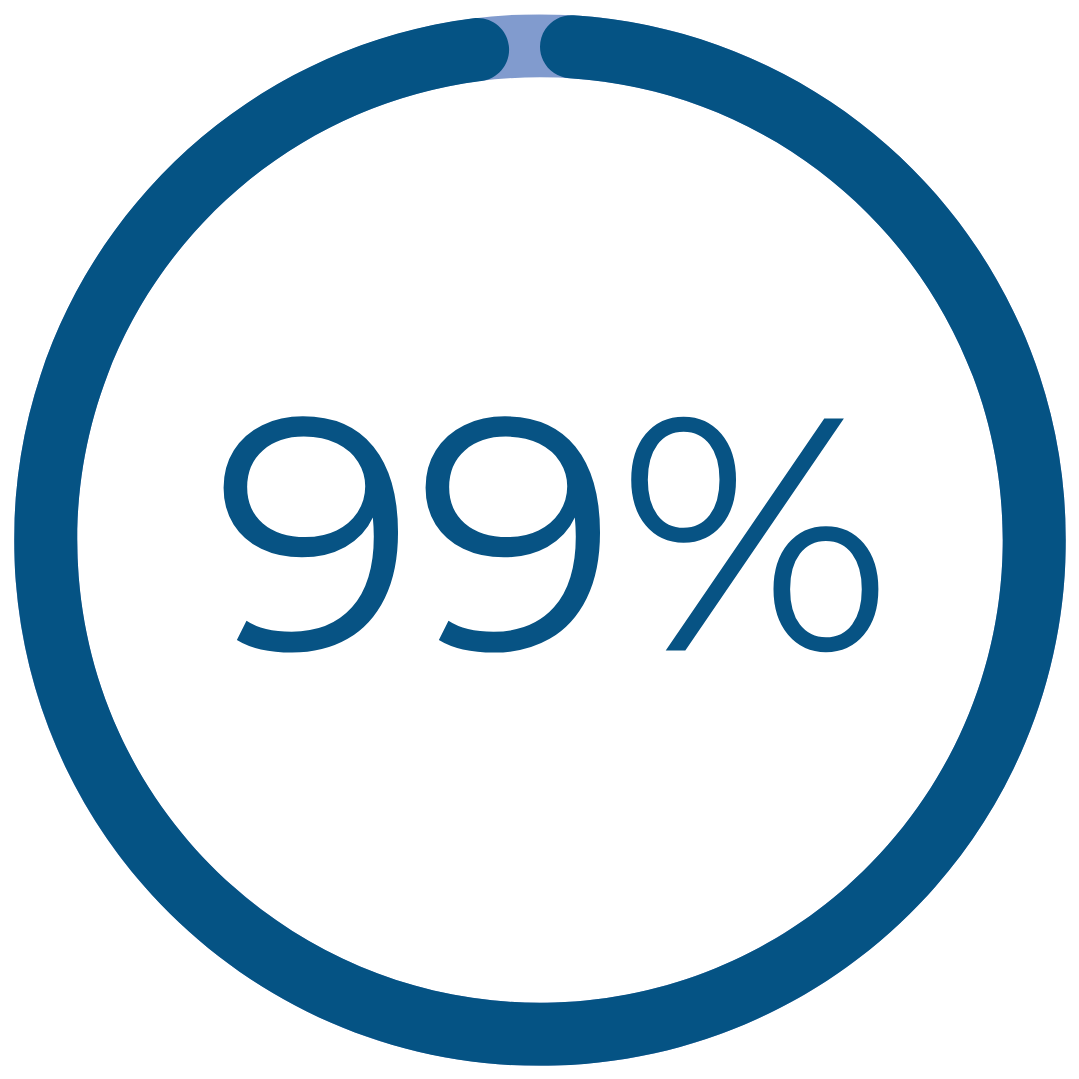
Results That Matter
reported normal, light or no periods†
had reduction in dysmenorrhea‡
would recommend Cerene to family and friends†††
uterine cavity entry with hysteroscope
† Patient-reported data are 1 year after treatment with durable results at 3 years.
‡ Improvement reported one year after treatment for patients reporting severe/very severe period pain.
††† Patient-reported data include definitely and consider recommending Cerene and are 1 year after treatment with durable results at 3 years.
What Your Peers Are Saying
About the
Cerene Device
Self-contained single-use system does not require capital equipment, so any room becomes a procedure room
Simple one-button device design, step-by-step guidance on the LCD screen, and a slim 6mm probe with flexible distal end that conforms to the uterine cavity
Radiopaque, thermally-insulating sheath facilitates accurate placement and visualization on ultrasound
Precise and automatically controlled treatment parameters with multi-tiered safety features
How it Works
2 1/2 minute cryotherapy treatment time
Gradual uterine pressurization and distention for patient comfort
Conveniently designed to fit into your practice routine